|
The gonad in mammal is comprised with two major cell types, germ cells and somatic cells. The primordial germ cells come from the extraembryonic ectoderm during early stage of embryonic development, whereas the somatic cells come from genital ridge. The sex determination in mammals is complicated process which is dependent on the differentiation of genital ridge somatic cells. In XX gonads, the somatic cells differentiate into granulosa cells and germ cells initiate meiosis right after sex commitment. By contrast, the somatic cells in XY gonads are developed into Sertoli cells and the germ cells will not start meiosis until after birth. The differentiation of genital ridge somatic cells is genetically regulated and many factors are involved in this process. Aberrant differentiation of gonad somatic cells will cause germ cell tumor, abnormal gonad development, and sex reversal. The research works in our group mainly focus on the regulation of somatic differentiation during sex determination and the interaction between germ cells and somatic cells in germ cell fate determination.

Wt1 is required for Sertoli cell lineage maintenance. Inactivation of Wt1 results in Sertoli cell-to-Leydig cell transdifferentiation, which in turn leads to the loss of Sertoli cell-specific gene expression and testicular cord disruption.
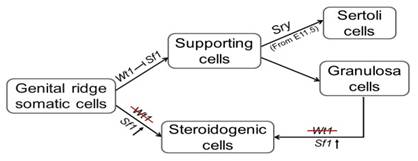
The supporting cells and a subset of some, if not all, steroidogenic cells (Leydig and theca-interstitial) are most likely derived from the same progenitor cells. Physiologically, these supporting cells will differentiate into Wt1-expressed granulosa or Sertoli cells in female and male gonads, respectively. By contrast, without Wt1 expression, the progenitor cells develop into steroidogenic cells and sex differentiation will not occur.
|
Plain English:
1. The regulation of supporting and steroidogenic cell differentiation during sex determination. Supporting cells (Sertoli and granulosa) and steroidogenic cells (Leydig and theca-interstitium) are two major somatic cell types in mammalian gonads. However, the mechanisms that control the differentiation between them during gonad development remain unclear. Our studies demonstrated for the first time that these two cell types are derived from same progenitors. We will explore the underlying mechanisms using gene knockout mouse models and in vitro system.
2. The regulation of germ cell meiosis initiation. In XX gonads, the germ cells initiate meiosis right after sex commitment, whereas the germ cells in XY gonad will not start meiosis until after birth. Several genes have been reported to play critical roles in this process. However, the detail mechanism is still unclear. We have generated several meiosis deficient gene knockout mice models and these models will be used to systematically study the regulatory mechanism of germ cell meiosis.
Selected publications:
- Zhang L, Chen M, Wen Q, Li Y, Wang Y, Wang Y, Qin Y, Cui X, Yang L, Huff V, *Gao F. Reprogramming of Sertoli cells to fetal-like Leydig cells by Wt1 ablation. Proc Natl Acad Sci U S A. 2015;112(13):4003-8.
- Wang Y, Zhu T, Li Q, Liu C, Han F, Chen M, Zhang L, Cui X, Qin Y, Bao S, *Gao F. Prmt5 is required for germ cell survival during spermatogenesis in mice. Scientific Reports. 2015; 5:11031.
- Gao F*§, Zhang J§, Wang XN, Yang JL, Chen DH, Huff V, Liu YX*. Wt1 functions in ovarian follicle development by regulating granulosa cell differentiation. Human Molecular Genetics. 2014, Jan 15;23(2):333-41.
- Wang XN, Li Z, Ren Y, Jiang T, Wang YQ, Chen M, Zhang J2, Hao JX, Wang YB, Sha RN, Huang Y, Liu X, Hu JC, Sun GQ, Li HG, Xiong CL, Xie J, Jiang ZM, Cai ZM, Wang J, Wang J, Huff V*, Gui YT*, Gao F*. The Wilms Tumor Gene, Wt1, Is Critical for Mouse Spermatogenesis via Regulation of Sertoli Cell Polarity and Is Associated with Non-Obstructive Azoospermia in Humans. PLoS Genetics. 2013, Volume 9, Issue 8, e1003645.
- Chen SR., Zheng QS., Zhang Y., Gao F*., Liu YX*. (2013) Disruption of genital ridge development causes aberrant primordial germ cell proliferation but does not affect their directional migration. BMC Biol. 11(1): 22. (*Co-corresponding author)
- Hu QH *., Gao F*., Tian WH., Ruteshouser C., Wang YQ., Lazar A., Stewart J., Strong LC., Behringer RR., and Huff V. (2011) Wt1 Ablation and Igf2-Up-regulation in Mice Result in Wilms Tumors with Activated pERK1/2. Journal of Clinical Investigation. 121(1): 174–183 (*Equal contribution)
- Chang H*., Gao F*., Guillou F., Taketo MM., Huff V., Behringer RR. (2008) Wt1 negatively regulates (beta)-Catenin Signaling during testis development. Development. 135(10):1875-1885. (*Equal contribution)
- Gao F., Maiti S., Alam N., Zhang Z., Deng JM., Behringer RR., Lécureuil C., Guillou F., Huff V. (2006) Wt1 is Required for Sox9 Expression and Normal Tubular Architecture in the Developing Testes. ProcNatlAcadSci U S A.103(32):11987-92
- Gao F., Maiti S., Sun GZ., Huff V. (2004) Mouse WT1 missense mutation results in glomerulosclerosis characteristic of human Denys-Drash Syndrome. Molecular Cellular Biology. 24(22):9899-910
- Gao F., Shi HY., Daughty C., Cella N., Zhang M. (2004) Maspin plays an essential role in early embryonic development. Development. 131:1479-1489

|

