| Brain is the organ that coordinates and controls body activities and functions, and serves as the center of memory, thought, and feeling. Neurogenesis, a process of generating functional neurons from neural stem cells and progenitor cells, is important for neuroplasticity and intricate neural network in the nervous system. The intricate neural network formation during embryonic and perinatal development is fundamental for proper brain functions. Aberrant neural network formation has been suggested to be the cellular basis for many mental disorders. We are interested in understanding the molecular and cellular mechanisms for stem cell maintenance and stem cell differentiation during early neural development and in adulthood. We are also interested in elucidating the roles of epigenetic genes in neurological diseases and mental disorders with neural developmental origins, such as autism. Finally, we would like to develop novel stem cell-based strategies to treat neurological diseases and nerve injuries. 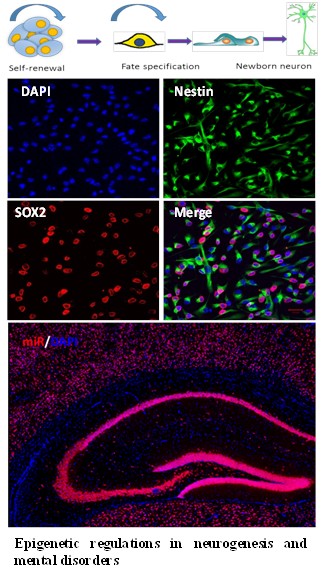
| Plain english:
Neurodevelopmental disorders are characterized by an abnormal development of the central nervous system (CNS), leading to learning and memory disabilities, attention deficits, speech disorders, mental retardation, or conduct disorders. Most neurodevelopmental disorders have complex and multiple contributors. Environmental risk factors, along with gene mutations, can affect neurodevelopment, including maternal use of alcohol, tobacco, or illicit drugs during pregnancy; and prenatal or childhood exposure to certain environmental contaminants. However, the pathogenic molecular mechanisms are not fully understood. A better understanding of the molecular pathogenesis will lead to developing novel therapeutic strategies for the treatment of neurodevelopmental disorders. Neural stem cells (NSCs) replacement and gene transfer have been considered as potentially powerful therapies for neurodevelopmental disorders. NSCs can differentiate into all kinds of neurons and glial cells, offering a unique and powerful tool for basic research and regenerative medicine. The focus of our research has been on identifying the epigenetic molecular mechanisms that might regulate cell fates of endogenous NSCs and on governing transplanted cells to accomplish a certain grade of morphological, anatomical and functional integration into the impaired host CNS tissue. We are also interested in generating functional cell subtypes from NSCs for cell transplantation and examining their safety and therapeutic potential for the treatment of neurodevelopmental disorders. Selected publications: - Ma JJ, Ju X, Xu RJ, Wang WH, Luo ZP, Liu CM, Zhou FQ, Saijilafu. Telomerase Reverse Transcriptase and p53 Regulate Mammalian Peripheral Nervous System and CNS Axon Regeneration Downstream of c-Myc. J Neurosci. 2019 Nov 13;39(46):9107-9118.
- Xi F, Xu RJ, Xu JH, Ma JJ, Wang WH, Liu CM, Yang HL, Meng B, Saijilafu. Calcium/calmodulin-dependent protein kinase II regulates mammalian axon growth by affecting F-actin length in growth cone. J Cell Physiol. 2019 Dec;234(12):23053-23065.
- Zhang SF, Gao J, Liu CM#. The Role of Non-Coding RNAs in Neurodevelopmental Disorders. Front Genet. 2019 Nov 20;10:1033.
- Yan HL, Sun XW, Wang ZM, Liu PP, Mi TW, Liu C, Wang YY, He XC, Du HZ, Liu CM#, Teng ZQ#. MiR-137 Deficiency Causes Anxiety-Like Behaviors in Mice. Front Mol Neurosci. 2019 Oct 30; 12:260.
- Xu YJ*, Liu PP*, Ng SC, Teng ZQ, Liu CM#. Regulatory Networks between Polycomb Complexes and Non-coding RNAs in Central Nervous System. J Mol Cell Biol. 2019 Jul 10. pii: mjz058 (*Co-first author).
- Liu C*, Dai SK*, Sun Z, Wang Z, Liu PP, Du HZ, Yu S, Liu CM#, Teng ZQ#. GA-binding protein GABPβ1 is required for the proliferation of neural stem/progenitor cells. Stem Cell Res. 2019 Aug; 39:101501(*Co-first author).
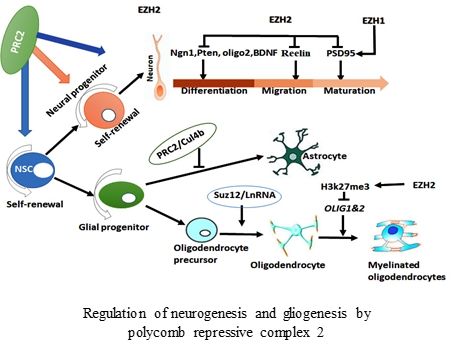
- Liu PP*, Xu YJ*, Dai SK, Du HZ, Wang YY, Li XG, Teng ZQ, Liu CM#. Polycomb Protein EED Regulates Neuronal Differentiation through Targeting SOX11 in Hippocampal Dentate Gyrus. Stem Cell Reports. 2019 Jul 9;13(1):115-131(*Co-first author).
- Cheng Y*, Wang ZM*, Tan W*, Wang X*, Li Y, Bai B, Li Y, Zhang SF, Yan HL, Chen ZL, Liu CM, Mi TW, Xia S, et al. Partial loss of psychiatric risk gene Mir137 in mice causes repetitive behavior and impairs sociability and learning via increased Pde10a. Nat Neurosci. 2018 Dec;21(12):1689-1703(*Co-first author).
- Duan RS*, Tang GB*, Du HZ*, Hu YW, Liu PP, Xu YJ, Zeng YQ, Zhang SF, Wang RY, Teng ZQ, Liu CM#. Polycomb protein family member CBX7 regulates intrinsic axon growth and regeneration. Cell Death and Differentiation. 2018 Sep;25(9):1598-1611(*Co-first author).
- Liu PP*, Xu YJ*, Teng ZQ, Liu CM#. Polycomb Repressive Complex 2: Emerging Roles in the Central Nervous System. Neuroscientist. 2018 Jun;24(3):208-220(*Co-first author).
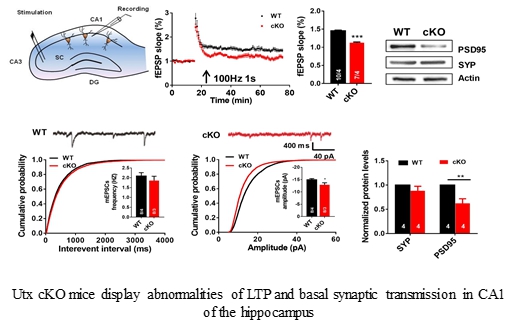
- Tang GB*, Zeng YQ*, Liu PP*, Mi TW, Zhang SF, Dai SK, Tang QY, Yang L, Xu YJ, Yan HL, Du HZ, Teng ZQ, Zhou FQ, Liu CM#. The histone h3k27 demethylase utx regulates synaptic plasticity and cognitive behaviors in mice. Front Mol Neurosci, 2017, 10:267(*Co-first author).
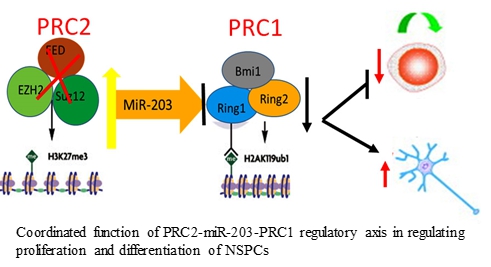
- Liu PP, Tang GB, Xu YJ, Zeng YQ, Zhang SF, Du HZ, Teng ZQ#, Liu CM#. MiR-203 Interplays with polycomb repressive complexes to regulate the proliferation of neural stem/progenitor cells. Stem Cell Reports. 2017,9(1):190-202.
- Li P, Teng ZQ, Liu CM#. Extrinsic and intrinsic regulation of axon regeneration by micrornas after spinal cord injury. Neural Plasticity, 2016:1279051.
- Jiang JJ*, Liu CM*, Zhang BY, Wang XW, Zhang M, Saijilafu, Zhang SR, Hall P, Hu YW, Zhou FQ. MicroRNA-26a supports mammalian axon regeneration in vivo by suppressing GSK3β expression. Cell Death and Disease. 2015, 6: e1865 (*Co-first author).
- Liu CM*, Wang RY*, Saijilafu, Jiao ZX, Zhang BY, Zhou FQ. MicroRNA-138 and SIRT1 form a mutual negative feedback loop to regulate mammalian axon regeneration. Genes Dev. 2013, 27(13):1473-83(*Co-first author).
- Liu CM*, Teng ZQ*, Santistevan NJ, Szulwach KE, Guo W, Jin P, Zhao X. Epigenetic regulation of miR-184 by MBD1 governs neural stem cell proliferation and differentiation. Cell Stem Cell. 2010, 6(5):433-44 (*Co-first author).
- Saijilafu, Hur EM, Liu CM, Jiao ZX, Xu WL, and Zhou F-Q. PI3K-GSK3 pathway regulates mammalian axon regeneration by induction of Smad1. Nature Communications, 2013, 4:2690.
- Guo W, Zhang Li, Christopher DM, Teng ZQ, Fausett SR, Liu CM, George OL, Klingensmith J, Jin P, Zhao X. RNA binding protein FXR2 regulates proliferation and differentiation of adult hippocampal neural stem cells by repressing Noggin expression. Neuron, 2011, 70:924-938.
- Liu CM, Teng ZQ, McQuate AL, Jobe EM, Christ CC, Hoyningen-Huene SJ, Reyes MD, Polich ED, Xing Y, Li Y, Guo W, Zhao X. An Epigenetic Feedback Regulatory Loop Involving MicroRNA-195 and MBD1 Governs Neural Stem Cell Differentiation PLoS One. 2013,8(1): e51436.
- Cong HL, Sun LQ, Liu C, Tien P. bition of atrogin-1/MAFbx expression by adenovirus-delivered small hairpin RNAs attenuates muscle atrophy in fasting mice. Hum Gene Ther. 2011 Mar;22(3):313-24.
- Li X, Barkho BZ, Luo Y, Smrt RD, Santistevan NJ, Liu C, Kuwabara T, Gage FH, Zhao X. Epigenetic regulation of the stem cell mitogen Fgf-2 by Mbd1 in adult neural stem/progenitor cells. J Biol Chem. 2008, 283 (41):27644-77652.
- Luo Y, Shan G, Guo W, Smrt RD, Johnson EB, Li X, Pfeiffer RL, Szulwach KE, Duan R, Barkho BZ, Li W, Liu C, Jin P, Zhao X. Fragile x mental retardation protein regulates proliferation and differentiation of adult neural stem/progenitor cells. PLoS Genet. 2010, 8;6(4): e1000898.
- Liu CM, Zhao XY. MicroRNAs in adult and embryonic neurogenesis. Neuromolecular Med. 2009,11 (3):141-152.
- Liu CM, Yang Z, Liu CW, Wang R, Tien P, Dale R, Sun LQ. Myostatin antisense RNA-mediated muscle growth in normal and cancer cachexia mice. Gene Ther. 2008, 15 (3):155-160.
- Gong H, Liu CM, Liu DP, Liang CC. The role of small RNAs in human diseases: potential troublemaker and therapeutic tools. Med Res Rev. 2005, 25(3):361-381.
- Ke XS, Liu CM, Liu DP, Liang CC. MicroRNAs: key participants in gene regulatory networks. Curr Opin Chem Biol. 2003 7(4):516-523.
|

